
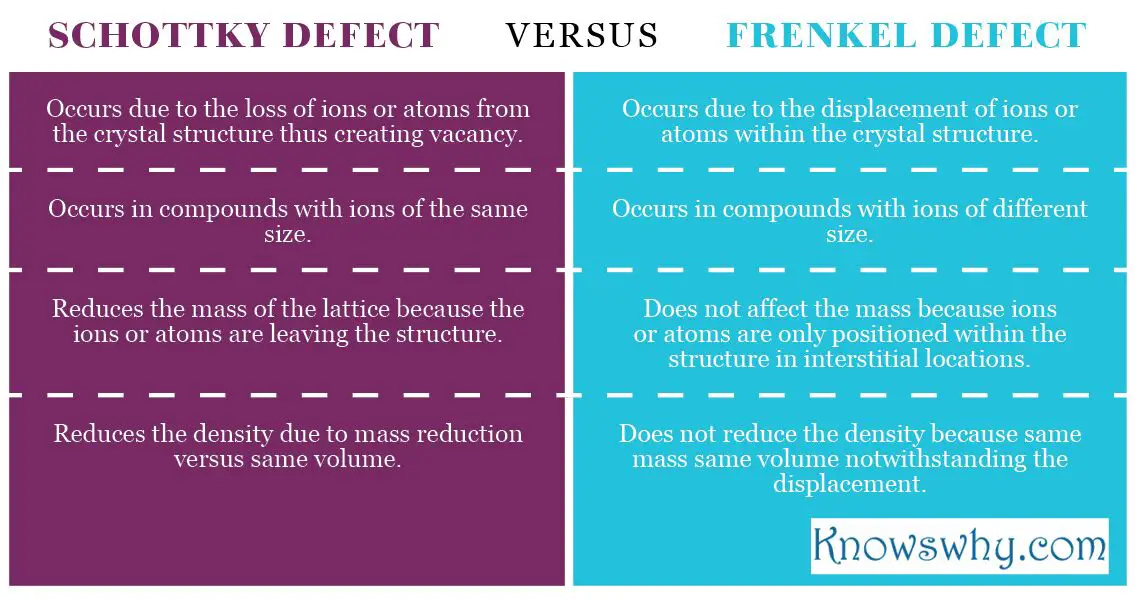
Lattice structures show often imperfections especially when their temperatures are greater than 0 Kelvin. The common defects are the point defects know as Schottky and Frenkel defects. Their names honour the first scientist that discovered these defects. Below are the differences between these defects.
Definition of Schottky defect
Schottky defect was named after Water H. Schottky who was the first person who discovered this point defect. The defect can occur in both the non-metal and metal compounds in which atoms or ions can leave their lattice sites thus forming vacancies in stoichiometric units. As they leave the lattice sites from stoichiometric units they still maintain the neutral charge and the balance of ions and atoms as reflected in the chemical formula.
As a result of these vacancies in schottky defect, the crystalline solid may experience a decrease in the density because the mass is being reduced while the volume remains the same. Normally, this defect occurs in crystal lattice with ions of equal size whereas the frenkel defect usually occurs in lattices with ions of different sizes. The compounds that often depict the schottky defect include NaCL, TiO2, KBr and KCL. When schottky defect occurs in non-ionic crystals, it is termed vacancy defect.
Definition of Frenkel defect
Frenkel defect on the other hand, occurs in crystal lattice where an atom or an ion is displaced from its original position to an interstitial position within the lattice. It creates a vacancy at an original position. This type of defect usually occurs in ions or atoms of different sizes.
As the ion or atom is being displaced within the lattice, the mass and the density remain unaffected in the same volume, so they remain constant, unlike with the schottky defect where atoms or ions can vacant the lattice completely. A frenkel defect will not alter the chemical properties of the compound. The defect was named after Moases Frenkel as the first person who discovered it in 1926.
Frenkel defect is evident in compounds such zinc sulphide and silver iodide. It happens mostly in ionic crystals, unlike the schottky defect that happens in both ionic and non-ionic crystals. The cation is usually smaller in these compounds that show frenkel defects mainly due to the increased effective nuclear charge.
Key differences between schottky defect and frenkel defect
Occurrence
- Schottky defect occurs in both ionic and non-ionic crystals and it is characterized by the vacancy of ions or atoms in the lattice. Usually, it occurs in the crystals with ions or atoms of the same size
- Frenkel defect occurs in ionic compounds where ions or atoms are being displaced from their original positions to interstitial locations within the lattice. Usually, it happens in compounds of ions or atoms with different sizes
Change in density
- Schottky defect leads to changes ibn the density of the crystal because the volume remains the same while the mass is reduced because ions or atoms have left the crystal and created the vacancies
- Frenkel defect has no effect in the density because the ions or atoms are just being displaced. So the mass remains the same and the density is unchanged.
Size of the ions
- Schottky defect occurs in ions and atoms of the same size
- Frenkel defect occurs in ions and atoms of different sizes
Chemical properties
- Schottky defect leads to a change in chemical properties of the compound
- Frenkel defect does not amount of changes in the chemical properties because the crystalline structure remains balanced because the atoms are only displaced
Summary
- Schottky and Frenkel defects are known as point defects
- Schottky defect is characterized by the vacancies of ions or atoms out of the lattice
- Frenkel defect is characterized by the displacement of ions or atoms within the crystal structure
- Schottky defect can occur in non-metallic and metallic compounds
- Frenkel defect occurs in ionic compounds
- Schottky defect changes the mass and consequently the density in a constant volume
- Frenkel defect does not affect the mass and the density
- Schotty defect usually happens in a lattice with ions or atoms of the same
- Frenkel defect occurs in lattice with ions of different size
Author: Lusi Madisha
Lusi is a professional writer and a researcher for more than seven years. She is a chemical engineering graduate from the University of South Africa.










Leave a Reply