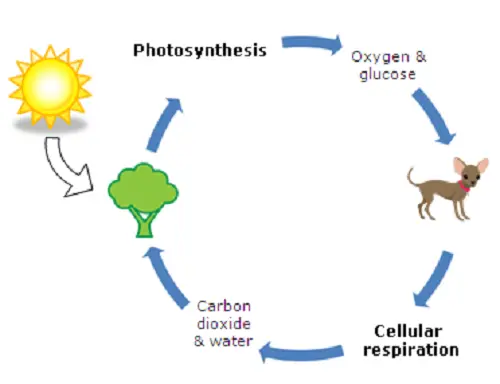
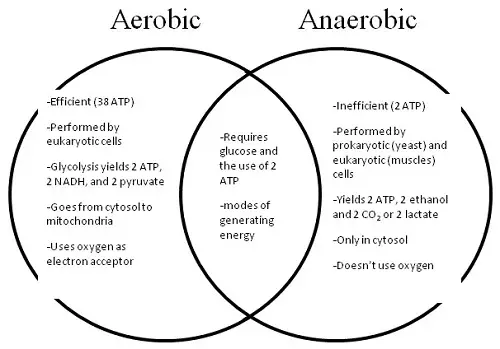
ATP, or adenosine triphosphate, is a molecule that literally contains energy for the various cellular processes in the body. Without the energy provided by the ATP molecule, cells wouldn’t be able to perform their functions efficiently. It is for this reason that ATP is referred to as a high-energy molecule. Its energy content basically fuels all the cellular processes in the body for proper functioning. Without ATP, the body will literally shut down and not function as expected.
The ATP molecule is produced inside the cells of all living things including human beings. Specifically, it is in the mitochondria where ATP is created to provide energy for the body. ATP in itself is not the energy source but, rather, the energy is stored in the three phosphate bonds of the molecule. When these phosphate bonds group together, it literally creates a spark that releases energy for the various cellular processes in the body. The process requires three bonds of phosphate to be able to release energy. At first, there may be only two bonds, and this is called ADP, or adenosine diphosphate. But when a third phosphate bond is created in the mitochondria, the stored energy is then released for use by the different cells of the body.
The best thing about this so-called high-energy molecule is that it is recyclable and re-usable by the body. Once the energy source is used up by different cellular or metabolic processes, all of the precursors to this molecule are converted back into ATP to provide a constant supply of energy source for the body. In this sense, the ATP molecule can be likened to rechargeable batteries that provide energy for various cellular and/or bodily functions when needed. The mitochondria in various cells continue creating the phosphate bonds to prepare and store energy for the body’s energy requirements.










Leave a Reply