The terminology nucleotides and nucleosides are often used when discussing the molecular and structural components of nucleic acids and they sound very similar.
Both nucleotides and nucleosides form the building blocks of nucleic acid containing compounds like DNA and RNA which are needed to form the structural components of cells. In addition, they are also needed to replenish cells and while both are actually classified as the same type of molecules, the small structural differences between them in turn affect the functioning of the individual molecules themselves.
The structure of nucleotides and nucleosides
The structure of nucleotides and nucleosides are quite similar and can easily be confused.
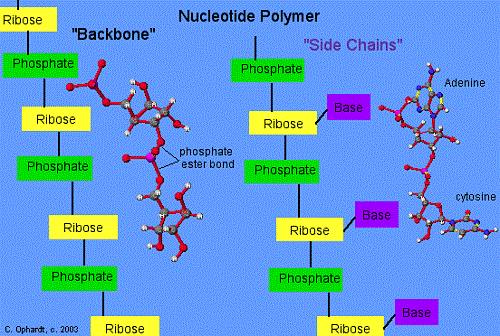
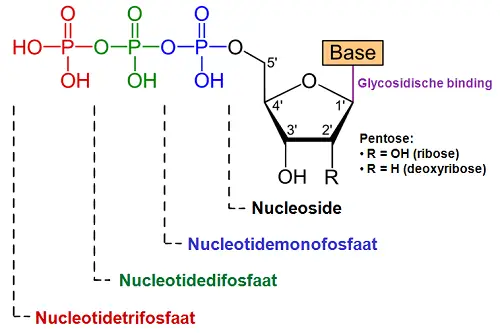
A Nucleotide is defined as a compound that consists of a nitrogenous base, a phosphate group and a pentose sugar. The pentose sugar can be attached from one to three different phosphate groups, and this sugar group can be either a ribose or deoxyribose sugar. The nitrogenous base can be either a purine or pyrimidine which differs based on their ring structure. Purines are classified by their double-ring structure while pyrimidines contain a single-ring structure. Purine bases include adenine and guanine while pyrimidine bases include cytosine, uracil and thymine.
A Nucleoside does not have a phosphate group bound to the 5’ carbon of the pentose sugar. The nitrogenous bases and the pentose sugar itself is the same as those found in nucleotides with the nitrogenous base being bound to the 3’ position of the pentose sugar. This bond occurs via a beta-glycosidic bond. Therefore it can be said that a nucleotide is a sugar + base + phosphate while a nucleoside is a sugar + base – phosphate. When nucleosides are phosphorylated by specific kinases, then nucleotides are produced.
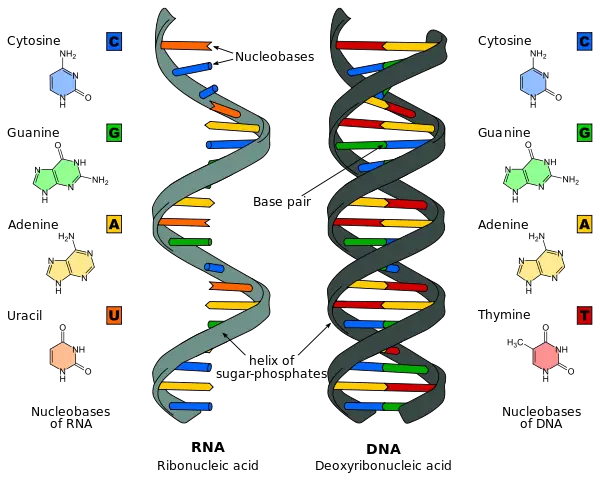
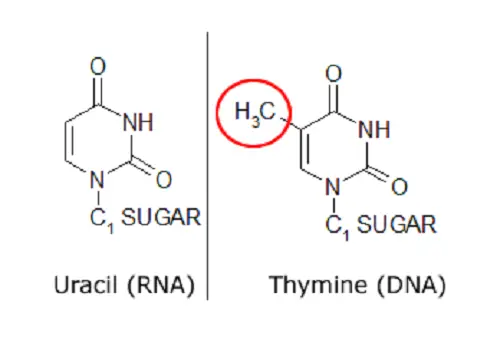
The polymerization of nucleotides leads to the formation of DNA and RNA molecules which connect from end to end to form a backbone-like structure via the removal of a diphosphate. This diphosphate removal allows the linking of the 5’ phosphate group of one nucleotide with the 3’OH group of another nucleotide through the formation of a phosphodiester bond. If ribose is the sugar within the nucleotide then this leads to the formation of RNA while deoxyribose leads to the formation of DNA. The nitrogenous bases found within RNA include adenine, guanine, cytosine and uracil while in DNA, the uracil base is replaced with thymine. Two strands of DNA can then be held together by hydrogen bonds which form between complimentary nucleotides as listed below:
Adenine – Thymine
Cytosine – Guanine
The two strands of DNA when hydrogen-bonded together forms what is known as a double stranded helical molecule.
Biological functions of Nucleotides and Nucleosides
Based on the above highlighted structural differences, it can then be said that both nucleotides and nucleosides will exhibit different biological functions within the body.
Nucleotides are known to form the basic building blocks of nucleic acids, DNA and RNA. These nucleotides are usually arranged into a chain that is linked by covalent bonds to form a sugar-phosphate backbone.
The four different types of nucleotides will repeat themselves along the chain in various patterns throughout the strand. These nucleotides can subsequently form longer chains which can then code for a host of amino acids. This coding will however depend on the arrangement of the nucleotides and the folding of the chain itself. This coding will in turn lead to the formation of protein structures.
Nucleotides are also involved in a host of biological functions and adaptations. These include data storage, energy holding, cellular communications and co-enzyme catalysis. Nucleotides analogues within the RNA sugar backbone may be used to regulate gene expression. In addition, they are also important for the proper functioning of the metabolism as well as maintenance and boosting of the immunity within the cell.
Nucleotides can function as a source of energy (also known as ATP or GTP) which can then be used in a range of biochemical processes or stored within the cell. It is also involved in the signal transduction pathways within the nervous system and endocrine system. Nucleotides such as Nicotinamide adenine dinucleotide phosphate (NAPD) and Flavin adenine diphosphate (FAD) act as cofactors in lipid and nucleic acid synthesis.
Nucleotides such as cyclic adenosine monophosphate (CAM) can also be used for signal transduction pathways within the cell.
Nucleosides can be used to form messenger RNA molecules (mRNA) which can subsequently copy information form a particular DNA strand. This copied information can then specify what information should be incorporated into amino acids and subsequently what type of proteins with form.
In addition to mRNA, nucleosides are also involved in the formation of ribosomal RNA (rRNA) and transfer RNA (tRNA). The main function of rRNA is to read the information on the amino acids and link them together. rRNA is also involved in the formation of new peptide bonds when proteins are being synthesized while transfer RNA is known to serve as an intermediate between amino acids and the mRNA.
The tRNA helps to decode the information from the messenger RNA. In addition, analogues of nucleosides are commonly used as antiviral and anticancer agents.
Conclusion
While nucleotides and nucleosides may seem similar in structure, the differences in arrangements of their components parts lead to a host of specific functions.
Nucleosides form the precursor molecules for nucleotides as they only lack a phosphate group while nucleotides on the other hand, form precursor molecules to both DNA and RNA molecules.
Despite this minor structural difference, the dissimilarities in the functions of these two groups are quite apparent leading to drastic changes within the cells.
Summary of differences between nucleotides and nucleosides










Leave a Reply