What is Valency?
The property of the atoms of an element to be chemically bonded to an exact number of atoms of other elements is called valency. Valency is an integer that shows how many hydrogen atoms can be joined or replaced by the atom of an element.
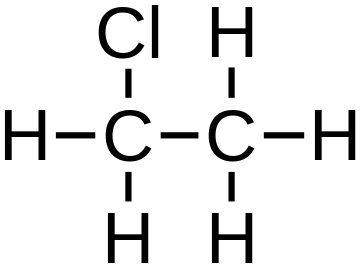
The valency depends on the ability of the atoms to:
- provide,
- accept,
- and share electrons.
From the point of view of the atomic construction theory, valency is an atomic property of a chemical element to participate in the formation of chemical bonds with a number of valence electrons. The numerical value of valency is determined by the number of electrons with which one atom participates in the generation of chemical bonds as a result of a chemical interaction.
Only electrons, located in the outermost shell of an atom can participate in a chemical reaction because they have very high energy. These electrons are called valence electrons and the valency of the chemical elements is equal to the number of these elections.
There are chemical elements, which do not combine with others. They are called inert or noble gases. The electron’s arrangement of the outermost shells of their atoms is very stable, which prevents them from chemical reactions. So valency can be determined also as the number of electrons, which the atoms of an element provide, accept, or share in order to achieve a stable inert gas configuration. As a rule, the valency of the chemical elements equals to:
- metals’ valency= number of the valence electrons,
- non-metals’ valency = 8 minus the number of valency electrons.
The valency of the elements can be:
- constant valency,
- variable valency.
The valency of a chemical element depends on:
- the state of its atoms,
- the nature of the other elements to which it is associated,
- the conditions under which the reaction occurred.
For example, in an oxygen deficiency, carbon forms the carbon oxide in which its valency is two, and in excess of oxygen – the carbon dioxide in which its valency is four.
In the cases, when the elements are providing or gaining electrons their valency can be:
- positive valency – for elements providing electrons,
- negative valency – for elements gaining electrons.
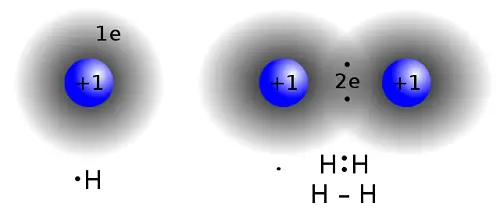
What is Covalency?
The number of bonds that can be formed by the atoms of a particular element by sharing its electrons with other atoms with empty orbitals is called covalency. The covalency is an integer and is always a positive number.
The total number of covalent bonds that the atoms of a certain element are capable to form is limited. The saturation of the covalent bond is due to the limited covalence capabilities of atoms, i.e. their ability to form a strictly defined number of bonds. The total number of valency orbitals in the atom (those that can be used to form chemical bonds), determines the maximum possible covalence of the element. The number of the already used for bonding orbitals determines the covalence of the element in a certain chemical compound. Thus, the covalence of an element is generally characterized by the total number of valence orbitals participating in the formation of covalent bonds.
The covalency of the elements can be:
- constant covalency,
- variable covalency.
Similarities Between Valency and Covalency
The similarities between the valency and the covalency are as follows:
- Valency and covalency are determined by the configuration of the atoms’ outermost shell.
- Valency and covalency are related to the ability of the elements to participate in chemical reactions and form chemical bonds.
- Valency and covalency are determined by the atoms’ seeking of the stability of the noble gas notation.
- Valency and covalency are constant for some elements and variable for others.
- Valency and covalency are integers,
Difference Between Valency and Covalency
1. Definition
Valency:
The property of the atoms of an element to be chemically bonded to an exact number of atoms of other elements is called valency. It shows how many hydrogen atoms can be joined or replaced by the atom of an element.
Covalency:
The number of bonds that can be formed by the atoms of a particular element by sharing its electrons with other atoms with empty orbitals is called covalency.
2. Numerical value
Valency:
The valency can be a positive or a negative integer.
Covalency:
The covalency is always a positive integer.
3. Bonding
Valency:
The valency pertains to elements with ionic and covalent bonds.
Covalency:
The covalency pertains to elements with covalent bonds.
4. Empty Orbitals
Valency:
Valency depends on the number of electrons, required for the filling of the available empty orbitals in an atom.
Covalency:
Covalency is determined by the number of empty orbitals in an atom.
5. Exchange of electrons
Valency:
The valency pertains to atoms which provide, accept or share electrons.
Covalency:
The covalency pertains to atoms which share electrons.
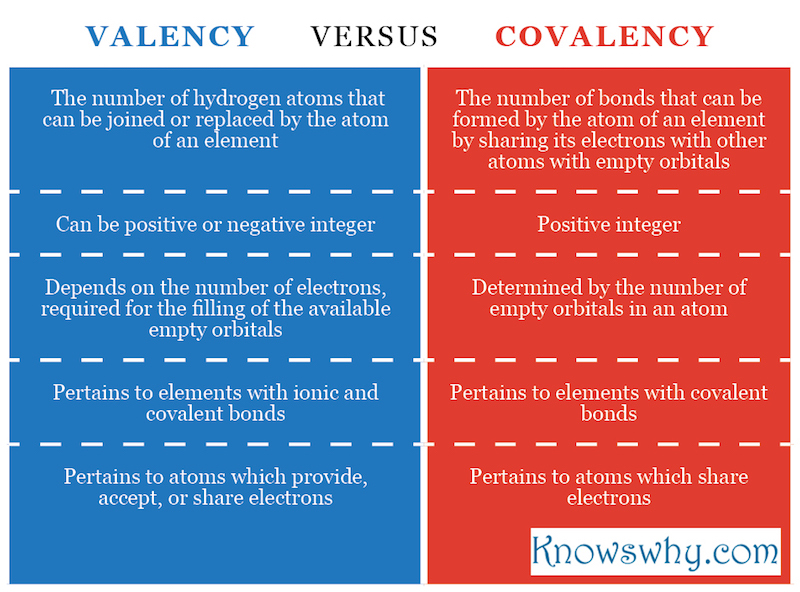
Summary:
- The property of the atoms of an element to be chemically bonded to an exact number of atoms of other elements is called valency. It shows how many hydrogen atoms can be joined or replaced by the atom of an element.
- The number of bonds that can be formed by the atoms of a particular element by sharing its electrons with other atoms with empty orbitals is called covalency.
- The valency can be a positive or a negative integer.
- It pertains to elements with ionic and covalent bonds and to atoms which provide, accept or share electrons.
- Valency depends on the number of electrons, required for the filling of the available empty orbitals in an atom.
- The covalency is always a positive integer.
- The covalency pertains to elements with covalent bonds and to atoms which share electrons.
- Covalency is determined by the number of empty orbitals in an atom.
Author: Dr. Mariam Bozhilova
Dr. Mariam Bozhilova has a Master’s degree in Ecology and PhD in Botany. Her main professional interests are in the fields of ecology, biology and chemistry. She has more than 10 years of professional experience in scientific research and environmental consultancy.










Leave a Reply