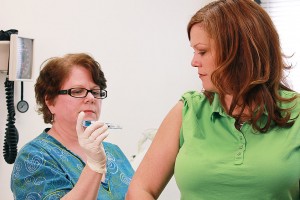
 Flu shots, or more properly influenza vaccines protect against the influenza virus. Typically, the vaccine consists of three or four vaccines targeting the predicated, by the World Health Organization (WHO), most common influenza strains for that season. As there is a southern and northern hemisphere season this means two vaccines are generated yearly. Due to the high mutation rate of the flu virus an individual vaccination usually only protects an individual for a few years. As this development of these vaccines can take several months, and it is not possible to vaccinate against all forms of flu at once, certain variants do get overlooked, or a new strain can arise during this time.
Flu shots, or more properly influenza vaccines protect against the influenza virus. Typically, the vaccine consists of three or four vaccines targeting the predicated, by the World Health Organization (WHO), most common influenza strains for that season. As there is a southern and northern hemisphere season this means two vaccines are generated yearly. Due to the high mutation rate of the flu virus an individual vaccination usually only protects an individual for a few years. As this development of these vaccines can take several months, and it is not possible to vaccinate against all forms of flu at once, certain variants do get overlooked, or a new strain can arise during this time.
Development
Following the devastating 1918 flu pandemic, sometimes referred to as the Spanish flu, there was widespread demand for the development of a vaccine. The first vaccine which received widespread use was developed by the US military and was used during and in the aftermath of World War II. This vaccine was generated in chicken eggs, a method which has remained most common although there are issues in those with egg allergies and in scaling up production to meet global demand. As such new methods of production are being developed including using vats of insect or animal cells which bypasses allergy issues and allows for bulk production1.
Method of Action and Delivery
Flu vaccines are available in two forms, both of which will target three or four viral strains. Most people will receive a vaccine generated in chicken eggs, although those at risk may be offered one of the newer egg free forms.
The traditional flu shots are delivered as a single injection into the muscle, usually of the upper arm, although variants which can be delivered under the skin or with a jet injector are available. The flu shot consists of an inactivated form of the influenza virus which means it cannot replicate, not infect new individuals. The recipient’s immune system detects the virus and generates an immune response. If at a later date they are re-infected with the real influenza virus their immune system will already be prepared to mount a response.
A newer flu vaccine is available as a nasal spray, making it easier to administer. However, this nasal spray vaccine uses a live attenuated virus, meaning that it is still capable of replication and or infection. Whilst this may not seem desirable live attenuated virus vaccines typically generate stronger immune responses which is of benefit following further infections.
Effectiveness
After delivery of either type of vaccine individuals will be protected from being infected by influenza, and if they do get infected the illness is much less severe with milder symptoms. Studies have shown that children (74%)2, adults (71%)3 and the elderly (77%)4 who received the flu shot were much less likely be hospitalized with flu symptoms, an even stronger effect was seen in pregnant women (92%)5. Perhaps more importantly reducing infectivity, illness duration and severity in individuals plays an important role in reducing the number of infected people in a population as a whole. It has been estimated that the flu vaccine program reduces overall numbers of flu cases by approximately 50%. However, as the vaccine does not protect against all flu strains, only those predicted to be most common, it does not provide complete protection.
Treatment Groups
The flu vaccine is recommended for all people with the exception of those with severe egg allergies. The WHO strongly recommends that high-risk groups such as pregnant women, children, the elderly, those with chronic illnesses and front line health care workers receive the vaccination6.
Potential Risks
Side effects of vaccination may occur, although they are usually minor and much less severe and common than complications arising from influenza infection. Those with severe egg allergies are not recommended to use the traditional vaccine and may be offered a more modern, egg free vaccination, those with milder allergies may be offered the standard vaccine and monitored7.
Guillain-Barré syndrome, a rare disorder whereby activation of the immune system leads to it destruction of the nervous system has been linked to the flu shot. However, the rates of occurrence vary considerably (1 in 1,000,000 to 1 in 10,000,000) and are significantly lower than the risk of influenza virus induced Guillain-Barré syndrome (1 in 10,000)8.










July 29, 2016 8:53 am
Hello You did not include the preservatives included in the vacines