Uncovering Chemical Bonds
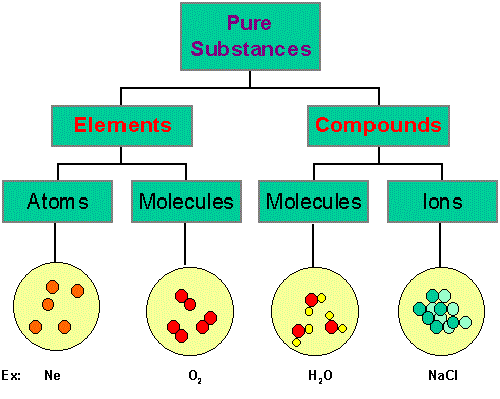
The attraction between atoms results into a chemical bond. These chemical bonds further bond with other atoms and/or molecules to form chemical compounds. Atoms that are not in a state of stability often attract other atoms and form a chemical bond. The occurrence of the bonding of atoms and molecules may occur naturally. Sometimes, several compounds are made to form chemical bonds with each other by scientists in the laboratory. The existence of these chemical bonds is extremely useful to us. The result of these chemical bonds are the substances that we use and need in our daily lives. These substances are also present in our body such as salt, sugar, and enzymes.
Why Do Chemical Bonds Form?
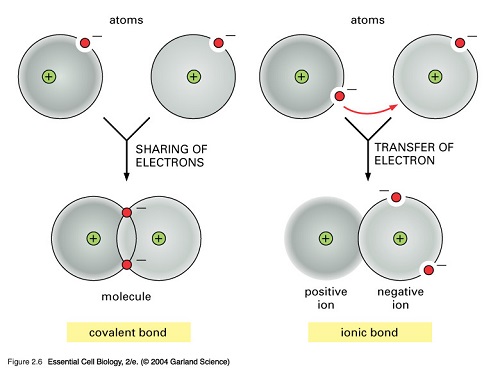
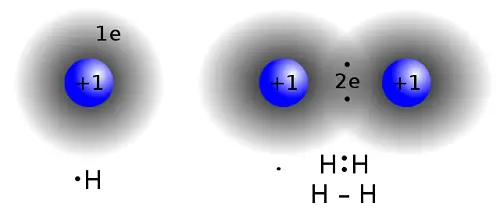
Chemical bonds occur to stabilize atoms. When the outer electron orbital of an atom lacks an electron, it seeks to bind itself to another atom such that its outer electron orbital will be filled. An atom always prefers its most stable configuration; thus, it forms chemical bonds with other atoms and rearranges until it is in its most stable form. The types of bonds that a molecule contains will be used to determine its physical properties, such as melting point, hardness, electrical and thermal conductivity, and solubility. In the formation of the chemical bonds, only the outermost or valence electrons are involved. The hydrogen, being the simplest element in the periodic table is composed of only two atoms. This element can be used as an example to show the formation of the chemical bond between the two atoms of the said element. When one hydrogen atom approaches another hydrogen atom, electron-electron and proton-proton repulsion both occur to prevent the two atoms from bonding. However, the electron-proton can counterbalance the repulsion, resulting in the formation of a chemical bond.
Chemical bonds occur in various ways. They depend on the type of element present and the activity between electrons that are involved in the bond. The three types of chemical bonds are ionic bond, covalent bond, and metallic bond. These chemical bonds formed result in different compounds which we often use in our lives.








Leave a Reply