
Why Do Ionic Compounds Dissolve in Water?
Water is considered to be a universal solvent meaning almost all compounds can be dissolved using it. Water is a polar molecule with a permanent dipole meaning it has a positive molecule on one side and a negative on the other. Hydrogen and oxygen molecules binding together form water. This is the reason why it can interact with almost all kinds of compounds because the oxygen has a negative charge and the hydrogen has a positive charge.
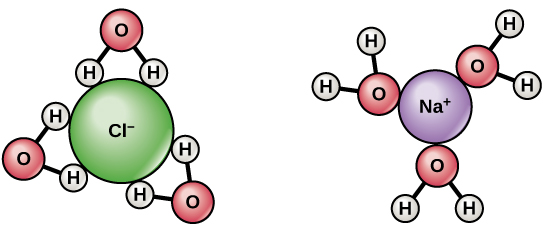
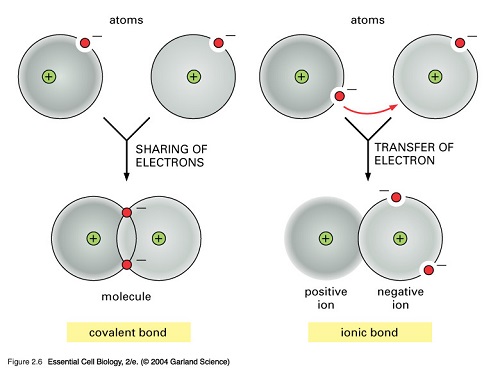
Ionic compounds on the other hand are formed when from the electrostatic attraction between oppositely charged ions. The electrons of an ion are transferred to another atom. The atom that loses an electron becomes a positively charged ion while the one that gains the electrons becomes the negatively charged ion. Salt or NaClis a crystal that is held together by an ionic bond and is the best example to use showing why ionic bonds dissolve in water.
Ionic compounds dissolve in the presence of water because the positive components of the compound are attracted to the negative charges. Then the negative charges of the compound are attracted to the positive charges of the water on the other hand. When this happens, the ionic bonds of the compound break as a result of the interaction between the water and the salt crystals. As the ions of the salt bind with the hydrogen and oxygen components of the water, each ion is surrounded by it creating a shell of polar water molecules with the broken ions from the salt in the middle.
The water acts as a stabilizer of the ions that are broken when the compound is dissolved. The water molecules surround the broken ionic components. Eventually when the water molecules separate all of the ionic compounds then you can say that it is fully dissolved by the water. The water that surrounds the broken up ions prevents the ions from getting together again.










Leave a Reply