
Why do Molecules Diffuse?
Diffusion is a process in which the molecules move from the area of high concentration of the substance to its low concentration. This happens with the help of the kinetic energy of the random movement of the molecules. If two boxes are filled with gas A in one and gas B in another separated by a partition, the molecules of the gases will collide with the partition and be in constant movement. If the partition is taken off, then the two gases will mix up with one another with their random velocities. After sometime, the gas molecules move in both directions and make a uniform concentration of A and B gas molecules all through the two boxes. The diffusion of molecules happens even at room temperature due to high velocities of the gas molecules.
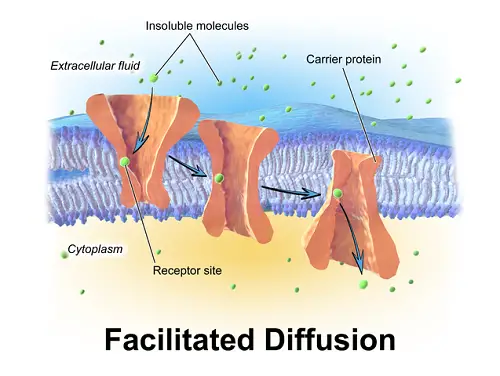
In diffusion, the solute will be totally and slowly spread all over the solvent. In the case of osmosis, the solvent molecules will move from their high concentration to low concentration through a semi permeable membrane. The molecules tend to diffuse because the solute molecules are attracted towards the solvent molecules and solute molecules repel each other. Hence, it is essential for the solute molecules to get distributed in the solvent equally.
The excitation of gas molecules is comparatively more than that of liquid molecules. This will result in repulsion of gas molecules amongst the gaseous substance. So, gases will get distributed or diffused fast in the solvent than the liquids. Diffusion is a passive process in the cell biology where the substances travel across the membranes. According to the second law of thermodynamics, entropy increases along with increase in disorder. Spontaneous processes result in dispersal of energy and they are irreversible. Diffusion is also a spontaneous process where the substance moves from its high chemical potential to its low chemical potential. This happens probably to increase the entropy or disorder in the molecules, repulsion between the electrons of the adjacent solute molecule and due to spontaneity in this process.









Leave a Reply