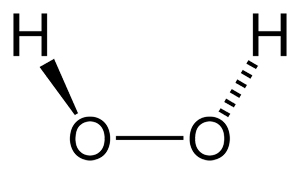
 Why does hydrogen peroxide bubble?
Why does hydrogen peroxide bubble?
The hydrogen peroxide that we get in a bottle from the store will not appear to be bubbling inside the bottle. As the concentration of hydrogen peroxide is only three percent in that bottle, it may not cause any intense reaction. Even when you put a drop or two on your skin, it will also will not cause any reaction or bubbles to occur. It is prescribed by physicians to clean wounds, as it works as an antiseptic. It is observed that H2O2 works similar to washing the wound with soap rather than acting as an antiseptic. But when hydrogen peroxide is poured onto a cut or wound it results in heavy foaming, and lots of bubbles are created.
The foam and bubbles are created when H2O2 reacts with blood and body cells that are present in the cuts and wounds. The blood and body cells consist of an enzyme called ‘Catalase’. This enzyme catalyzes the reaction 2 H2O2 —ÃÂ 2 H2O + O2. This reaction gives off lots of oxygen, as this reaction occurs about 200,000 times in one second. As oxygen is known to have healing capabilities, it is said that H2O2 cleans the wound. The heavy foam and bubble formation is due to the formation of oxygen. The same thing can be seen even when we put H2O2 on a cut potato.
There are other examples of bubbling hydrogen peroxide. On a rough surface it bubbles heavily, and on smooth surfaces it remains still. When earrings are put into a hydrogen peroxide liquid, it bubbles for some time and then stops. As the surface area is more rough, it tends to react more with the peroxide. Earrings might have either dust or dead cells on the, and hence cause bubbling. It is evident that rough surfaces and dust can act as catalyst of the above reaction. If there is any oxidized metal present in the earrings, then there is chance of it getting reduced by the hydrogen peroxide. Hence, it bubbles.










Leave a Reply