
Why Does Salt make water Boil faster?
Adding salt to water doesn’t make it boil faster, it makes it hotter. The boiling point of water is at 100 degrees celsius. If you add salt to water it will ‘increase, its boiling point from around 100-106. Adding salt to water takes it little longer to boil (but not too much, usually the intervals are just in a few seconds).
This process is best used when cooking. Adding salt to the water will make sure that the ingredients are cooked thoroughly and not to mention ‘faster,. That is all thanks to the increase in water temperature. According to a few experts, salt actually changes the chemicals found in water. You might notice that if you add salt to already boiling water, the surface would seem to get hotter and more ‘gaseous, for a brief period of time. Boiling doesn’t start under, but on the surface of the water.
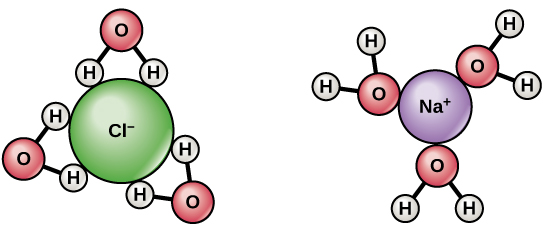
The chemicals found on salt (sodium chloride) can help change the chemical structure of the water molecules. The positive ions of salt will attract the negative ions of water and gas is released in the process. This process will help to expand and increase the rapid movement of the ions in the water, which in turn will yield to an increase in temperature.
People might have noticed this sudden change in the water’s temperature, so they say that when you add salt in water it boils faster. The ‘real truth, however, is that when you add salt to already ‘boiling water, it would appear to ‘boil even more,. This is probably the reason why people misunderstood, that salt actually makes the boiling process faster.
So the next time you cook, make sure to add salt in the water. It doesn’t only make the food taste good, it also helps in making it cook faster.










July 29, 2011 5:35 pm
This is not true. There’s no such thing as a salt molecule, it’s an ionic bond, so in water it just separates into the ions.
February 8, 2012 2:13 pm
“The positive molecules of salt will attract the negative molecules of water and gas is released in the process”. What?
October 20, 2014 11:11 am
Well you get the point of what he really means but you are right he should have said ionic bond.
April 22, 2012 1:06 am
I just have one question: if you add MORE salt to water than ever – which is at the boiling point of 106 degrees – will it become more hot and faster?