 Why is NF3 Polar?
Why is NF3 Polar?
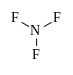
NF3 is the chemical representation for the compound nitrogen trifluoride. This inorganic compound possesses characteristics of being colorless, toxic, odorless, and nonflammable gas substance. Nitrogen trifluoride is most commonly used in the cleaning of plasma or thermal cleaning of chemical vapor deposition, also known as CVD, which is a chemical process that is used to produce solid materials of high performance and high quality. Aside from that, it is also used in processing plasma in order to create integrated circuits, which are emanated from electronic circuits, only smaller in size. Another use of nitrogen trifluoride is in chemical lasers, since it has handling convenient properties as well as it provides stability compared to fluorine gas.
As a greenhouse gas and with a global warming potential, nitrogen trifluoride is found to be a potent yet slow oxidizer. This compound has the same molecular structure with NH3 or ammonia. Nitrogen trifluoride has a tetrahedral or trigonal pyramidal shape. Because the compound is basically composed of a single nitrogen atom and three hydrogen molecules, there exists one lone pair of electrons. Since there are extra available electrons on the nitrogen atom, this makes the compound polar. Polarity is the ability of electrons to share and receive electrons whereby in the sharing of electrons, an unequal distribution among electrons is evident. When this happens, the molecular structure of the compound becomes asymmetrical. This is what is seen in the molecular structure of nitrogen trifluoride. Thus, this compound is deemed to be in a polar state.
Nitrogen trifluoride has found to have minor effects only when it comes in contact with the skin. However, it can cause severe reactions when inhaled and was found to be a pulmonary irritant that it can lead to toxicity when exposed severely to the substance.












Leave a Reply