
Why Water is considered a polar molecule?
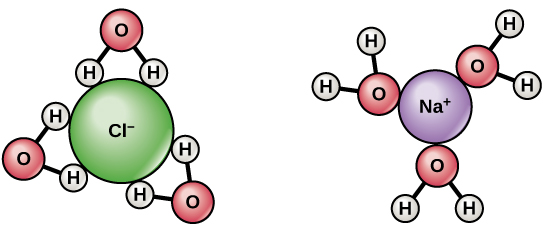
Water molecule is Polar due to its shape. Water has two positively charged hydrogen atoms and one negatively charged oxygen atom bonded covalently in a single water molecule. The hydrogen atoms have one electron in each of them and the oxygen has 4 electron pairs. Two electron pairs of oxygen form covalent bonding with two electrons of hydrogen. The remaining two lone pairs of electrons are actually responsible for the electro-negativity of oxygen. Due to the two lone pairs of electrons on oxygen atom, the shape of the water molecule appears as angular and the bond angle is obtained as 104.5 degrees. The bond angle for water will exist as slightly less than the tetrahedral angle. Hence, the molecular structure of water appears as angular.
The polarity of water is obtained when the oxygen tends to drag the shared electrons in the covalent bonding towards its side. Though in covalent bonding electrons are equally shared between the atoms, in the case of water molecule, the electrons are attracted strongly towards oxygen. This pulling of electrons by oxygen gives an asymmetrical distribution of charge. The asymmetry in the molecular structure offered a partial negative and partial positive charge. This polarity of water allows other substances to get dissolved into the water easily.
The polarity in water helps its molecules to remain close to one another by a force called cohesion. Polarity aids water molecules to get attached to other molecules with another force called adhesion. Due to this adhesive forces water tends to rise up in a tube making use of capillary action. The properties of cohesion and adhesion possessed by the water molecules help it to move from the roots to leaves in the xylem vessels. Water polarity is significant for animals too as they depend on plants for many purposes.
Water polarity helps it to act as universal solvent while the hydrogen bonding in between the water molecules exist due to the polar nature of hydrogen of one molecule and oxygen of another molecule.











Leave a Reply