Why does water boil?
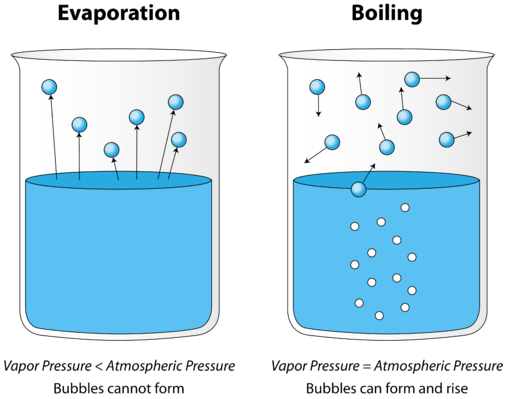
At normal or ordinary temperatures, water evaporates when left in the open air, although very slowly. The vapor formed from it has some pressure which is lower than the surrounding atmospheric pressure. The water also tries to form bubbles which are mixed up with the air. At low temperatures, they burst because the pressure inside the bubbles is low, and the surrounding higher pressure breaks it down. When water is heated, then the pressure of its vapor reaches equal to that of the surrounding air, at 100 degrees Celsius. Even the bubbles that are formed inside the water are also not broken, as they possess air with high pressure. They rise up and reach the water surface, and release the air inside into the air.
At very high places, like on the mountain tops which are a few thousand feet high above sea level, the atmospheric pressure will be low. The water, or any liquid, needs less temperature to expand into gas. While on low grounds it needs more temperature to equalize its vapor with the surrounding air. As the outside pressure is low at heights, water can quickly reach that pressure level where it is evaporated.
Water boils to change its liquid state to a gaseous state, which is called water vapor. When water changes from a liquid form to a vapor form it takes up some energy. This energy is called the latent heat of evaporation. As the energy is provided as a temperature, it exists at 100 degrees Celsius only until all the water changes into vapor.
Water boils to make the pressure in the bubbles that are formed by the application of heat equalize with the atmospheric pressure. Water makes this happen at its boiling point. This boiling point of water differs based on the height at which the water must vaporize. At sea level, water requires a temperature of 100 degrees Celsius to make its vapor pressure reach the atmospheric pressure. As the height increases, lower temperatures can make the vapor pressure reach the atmospheric pressure.










April 16, 2014 1:18 am
It is not higher elevation that causes water to boil at lower temperatures, but rather decreasing barometric pressure. Thus, water at sea level will boil at different temperature depending upon whether it is in a high pressure area or a low pressure area.