
Why is Air a mixture?
Air is a mixture and not a compound as freezing of air forms different liquids at different temperatures. For example liquid nitrogen has different boiling point than the liquid oxygen. The freezing point of various components of the air varies. If the air has only one compound, then the air might have had one boiling point and one freezing point. The components of air are oxygen, carbon-di-oxide, and nitrogen which are present in the air in separate proportions. The components in the air are not changed by reacting with each other. Air is made up of 78.09 percent of nitrogen, 20.95 percent of oxygen, and the remaining part is made of 0.93 percent of argon, 0.03 percent of carbon-di-oxide. Air also consists of water vapor.
Element is something that cannot be separated into two components as all the atoms in it are of same kind. Hence, an element cannot be divided into simple substances. Air comprises of various elements that are not combined chemically. But, compounds are those which are combination of two or more elements that are bonded chemically. So, it can be said that air is a mixture of compounds and elements. Different percentages of individual compounds mixed in its own quantities are observed to be present in the air.
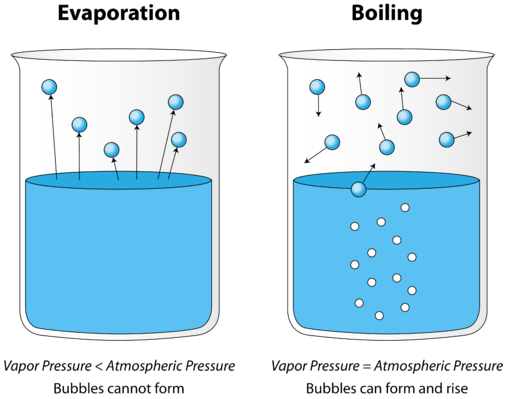
Air is considered as a mixture because it is composed of various separate components. The air has no characteristic properties of its own. The components of the air can be separated individually. Air cannot be indicated by a chemical formula. The boiling point of the air cannot be fixed. Air is otherwise called as homogeneous mixture or solution. One way to separate the components is to cool the air to a liquid and allow the different components to boil of. There are certain machines that separate air partially. As the components of the air are not chemically bonded with each other, air is considered as a mixture.











November 13, 2013 4:51 pm
i love science i would like to be a scienctist and an astronaut