
Why is Boiling point important?
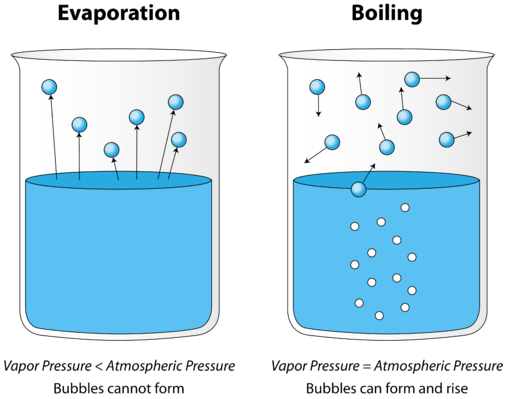
Boiling point is one of the physical properties of the chemical compound. Each chemical compound will have a specific boiling point. It is the temperature at which the liquid changes into gaseous form at standard atmospheric pressure. Boiling point can also be defined as the temperature at which the vapor pressure of the liquid is same as that of the external pressure. The normal standard boiling point is the temperature at which the liquid boils when the external pressure is 1 atmosphere.
It is evident now that the boiling point is dependent on atmospheric pressure. The boiling point becomes less when there is decrease in external pressure. At the boiling point, the extra heat added to the liquid converts it into vapor at the same temperature. It can also be said that at the boiling point the temperature and pressure of liquid and gas are in equilibrium.
Boiling point can be useful in determining whether the material is pure or not. If the melting point of any pure compound is known, then the melting point of the substance you have can be matched with the pure one. If the melting points are same it indicates that the substance you have is the same as the one that was compared with. If the boiling points are different, it indicates that the two substances are not same in their purity. The boiling point indicates the pressure of the external environment. The environment has high pressure on the liquid with high boiling point compared to the liquid that exists in atmospheric pressure. Boiling point provides the heat of vaporization that is used to alter the phase of saturated liquid.
Saturation temperature is the temperature at which the existing pressure is saturated. At the saturated pressure, the liquid boils to form vapors. The liquid will be saturated with the thermal energy which is actually responsible for conversion of phase.










Leave a Reply