
Why Are Hydrocarbons Hydrophobic?
Hydrophobic literally means the ‘fear of water,. Hydrocarbons are compounds that are made up of hydrogen and carbon molecules. Hydrocarbons have the ability to bond themselves together and form chains that holds different properties that repels water molecules.
One good example of hydrocarbon is oil. Oil and water don’t mix and would be forever separated from each other. The hydrophobic effect is caused by the hydrocarbons tendency to repel water molecules. The molecular structure of hydrocarbons creates a cage that doesn’t allow water to pass through them. An interaction between the molecules is limited and doesn’t encourage any progress with their molecular structures.
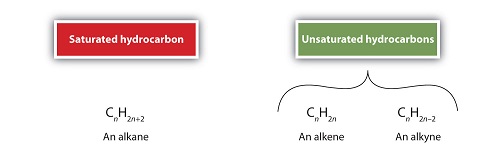
Hydrocarbons are non polar by nature and will naturally repel polar water molecules. This is where the hydrophobic effect got its name. There are many types of hydrocarbons and they are classified into saturated hydrocarbons, unsaturated hydrocarbons, cycloalkanes and aromatic hydrocarbons.
The importance of hydrocarbons lies in their ability to generate energy and is considered a valuable resource. Hydrocarbons have many forms and it has a solid, liquid and gas form. An example of the solid form of hydrocarbons is asphalt. Petroleum is one of the liquid forms of hydrocarbon. Hydrocarbons in general are a very important source of energy fuel and are used worldwide.
Fossil fuels and other forms of hydrocarbon mixtures allow the continuous growth and production of technology. Everything mechanical and electrical needs a constant source of energy. Without that source, the function of those equipments will come to a halt.
The use of hydrocarbons as fuels also affects the ozone layer. High concentrations of burned hydrocarbons can be dangerous to the health.










Leave a Reply