 Why Is NaCl Neutral?
Why Is NaCl Neutral?
NaCl or sodium chloride or better known as salt is considered as neutral in term of its components. In chemistry, a compound is considered neutral if it can’t be a base or an acid or it has a ph level of 7.
Aside from salt, water is also a neutral compound. But what makes these solutions neutral? Let’s take a look with its atomic components. NaCl is composed of Sodium (Na) and Chlorine (Cl). When these two elements are combined they are called Sodium Chloride. Sodium has a valency of +1 while Chlorine has -1. When these two form abound, there will be 0 valency. Valency is defined as the number of hydrogen atoms an element can combine with.
If we try to analyze the neutralization process, we will understand why salt is neutral. To simplify the explanation, all acids release a H+ while the bases release OH-. The reason why water is neutral is because when H+ and OH- are formed it will result to H20. One negative hydrogen plus one positive hydrogen equal 2 neutral hydrogen.
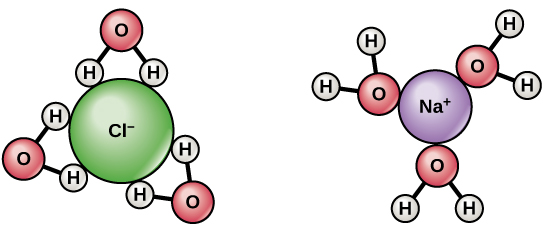
For salt or NaCl, this is what happens. When Na+ is combined with Cl-, it will become positive plus negative, therefore it will result to neutrality. Because of its Na+ characteristics, the sodium atoms will each lose an electron because it will be combined to an element without an electron and that is the Chlorine. That is why these two elements are always attracted to each other because they perform the give and take relationship. The sodium gives and electron while the chlorine takes the electron. When formed they become a sodium chloride which is a neutral ionic solution.
This also happens when a base and acid are formed together. The Acid (HCl) plus the base (NaOH) when combined will produce water and salt which are all neutral.









Leave a Reply