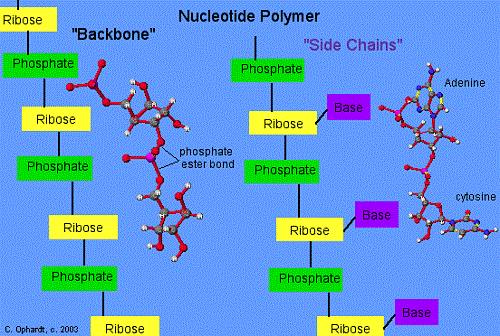
Genetic materials such as DNA and RNA are the language of life. All of the world’s living organisms are imbued with a blueprint for their creation. And that blueprint is written in the language of either DNA or RNA. Purines and pyrimidines form the building block for the making of DNA (also known as deoxyribonucleic acid).
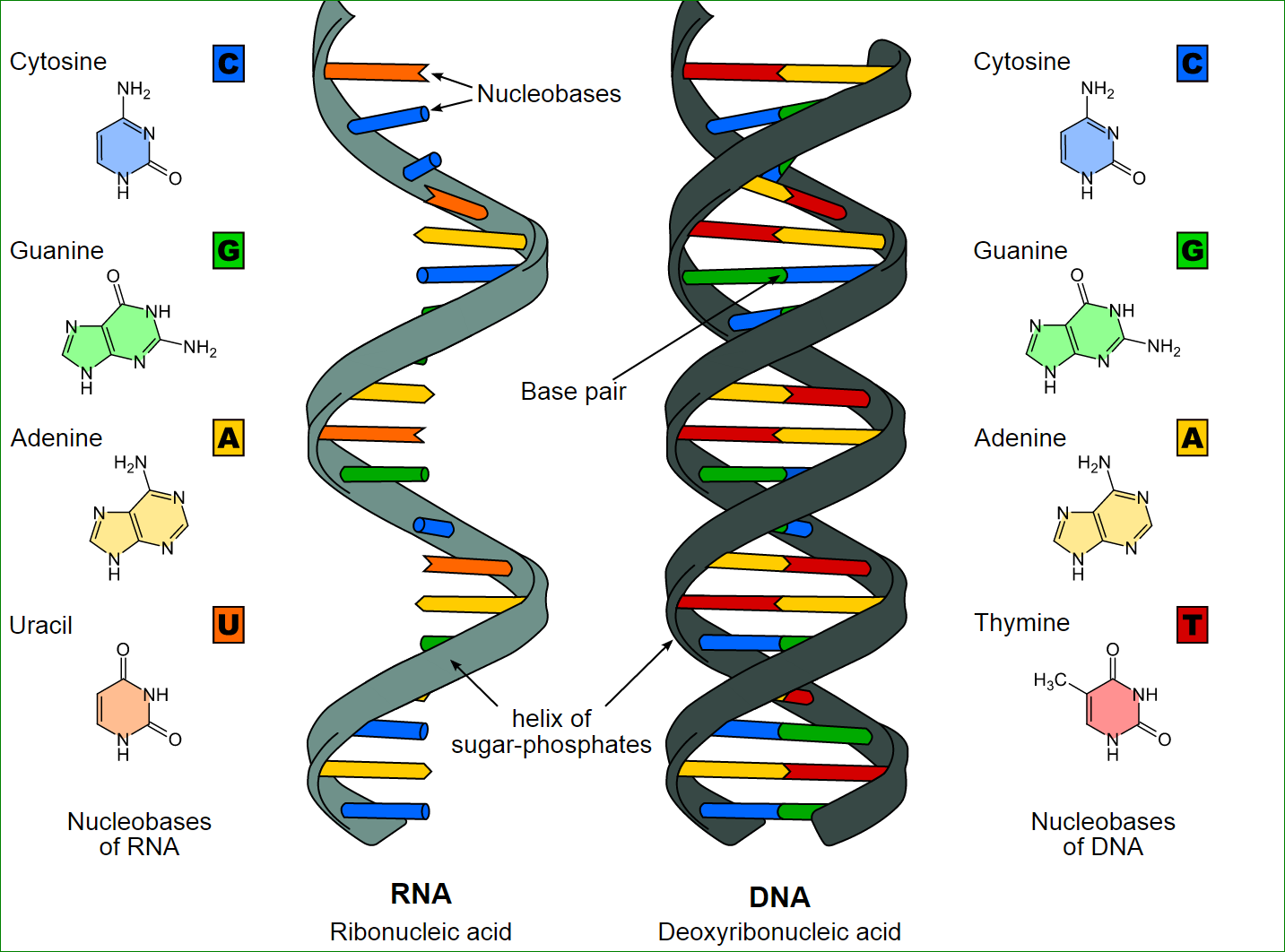
The structure of genetic material is so basic that there are only two purines and three pyrimidines in total. That’s right, just five chemicals that make up the entire genome of every single organism that exists today. Â Purines are bigger, with two ‘rings’ of base, and pyrimidines are smaller, with only one ‘ring’. The two purines are adenine and guanine, and the three pyrimidines are cytosine, thymine and uracil. Uracil is present only in RNA.
The structure of DNA is called a double-helix, and the best way to think of that is to imagine a ladder that’s been twisted like a spiral. The sides of the ladder are made of purines and pyrimidines. Each rung of the ladder is a hydrogen bond between a purine and a pyrimidine.
But why do purines only bond with pyrimidines? Why aren’t there bonds between two purines, or bonds between two pyrimidines? There are two reasons for this. The first reason is that the distance between the two strands of DNA is constant. Two purines together would be too big to fit in that distance, and two pyrimidines together would be too small.
The second reason is that the bonding between purines and pyrimidines is very specific. Not only can a purine not bond with anything but a pyrimidine, but specific purines have to bond with specific pyrimidines. This is because the structure of each is such that it can only bond with the corresponding one. The bonding rules are “ adenine with thymine, and guanine with cytosine.
This specific bonding pattern or rule is called Chargaff’s rule, after the scientist who discovered it. So specific bonding rules, secondary to chemical structure, and a constant distance between the two spirals, are the reason why purines only bond with pyrimidines.









Leave a Reply